Host specificity II:
host switching by viruses
Most of the examples I mentioned in class on Monday,
and all of the examples in the Poulin book, are
about "classical" metazoan parasites.
This topic covers host-switching in viral populations, continuing the
topic of host specificity and switching at many different evolutionary
levels.
Morbilliviruses
Biology
The morbilliviruses are part of the paramyxovirus group:
these are single-stranded RNA viruses, which use host biochemical
machinery to synthesize DNA and proteins from their RNA.
Within hosts: immunosuppressive, can be highly
virulent (esp. in inexperienced populations). However, recovery
generally brings lifelong immunity.
In general morbilliviruses
have a surprisingly low mutation rate (surprising because RNA copying
is messy, and because single-stranded viruses don't provide a
"reference copy" for fixing mistakes); they don't tend to
escape by genetic change in some of the ways that (e.g.) influenza and
AIDS do.
Since they typically lead
quickly to either recovery and death,
morbilliviruses often need large populations to sustain themselves
through a continuous flow of new susceptible hosts. Otherwise they
can "burn out" local populations (measles needs a host population of
about 250,000 to persist over long periods of time).
Morbilliviruses do not tend to have biochemical mechanisms for
persisting over a long period in a particular host individual,
although there are possible exceptions.
Since morbilliviruses "live fast and die young" both within hosts and
(of necessity) within host populations, they are always perched on the
brink of extinction. Interestingly, morbilliviruses as a whole have a
wide host range, and some viruses within the family have wide host
ranges. Is this a "solution" to the ecological problem that
morbilliviruses face?
Examples
- Measles virus (MV) is a fairly host-specific
virus, which infects humans and closely related primates
(although population structure within wild primates
doesn't allow persistence: critical community size is 300K
for humans).
Inexperienced human populations experience high mortality,
which could suggest some population specificity, but this probably has
more to do with host resistance than with parasite variation.
(Also, host mortality is not necessarily correlated with parasite
fitness: we'll talk about this at much greater length when we get to
discussing virulence.)
- Rinderpest (RPV): infects bovids, both domestic and wild.
Native to central Asia (which is a center of endemism for many
different taxa, including diseases), it did sometimes spread into
Europe
but didn't cross the Sahara until the 19th century.
Carried with livestock of an Italian expeditionary force,
it spread through sub-Saharan Africa in 10 years,
with huge ecological, social, and historical
consequences (which we may discuss more
later, when we get to "community and ecosystem consequences of
parasites").
For our purposes, the interesting thing is that rinderpest
affects both domestic bovids and a wide spectrum of wild
species.
- Peste des petits ruminants (PPRV/"goat plague"):
original source in West Africa, went the other way (now endemic in
mideast and southern Asia).
similar to rinderpest, affects goats and sheep and a
range of wild hosts
- Phocine & cetacean distemper viruses: there have
been recent discovery of a whole batch of these viruses in
causing marine mammal die-offs of various kinds: these
epidemics are really hard to observe, but bodies do wash up
(North Sea phocine distemper virus epidemic in 1988 is probably the
most carefully recorded example), and reverse transcriptase PCR
(RT-PCR) techniques are helping a lot because one can extract virus
from decomposing/ed samples (and sequence the virus, if
there's enough left, to find out phylogenetic relationships)
- Canine distemper virus: domestic dogs (obviously), also
affects wild dogs, hyaenas, and almost every other carnivore you can think of
(polar bears, raccoons, pandas, seals, ferrets, lions, etc. etc. etc.).
Responsible for many outbreaks in small and endangered wildlife
populations: e.g. blackfooted ferrets, lions of the Ngorongo crater
(30% (?) mortality in 1994).
Often spread from domestic dogs (which often have high, stable
population densities) but also (?) in some cases apparently
circulating/persistent within wild populations (polar bears, crabeater
seals).
Q: could patterns of host specificity
be related to population ecology of the host taxa?
(e.g. large, stable populations allow more specialized parasites?)
Host shifting
Morbilliviruses apparently switch hosts a lot.
In particular, we
can see shifts in specificity at three levels,
each of which has molecular phylogenies to back it up.
- the most "macro" level, the distinction between
different large branches of the phylogeny
(measles, rinderpest/PPRV, CDV/PDV)
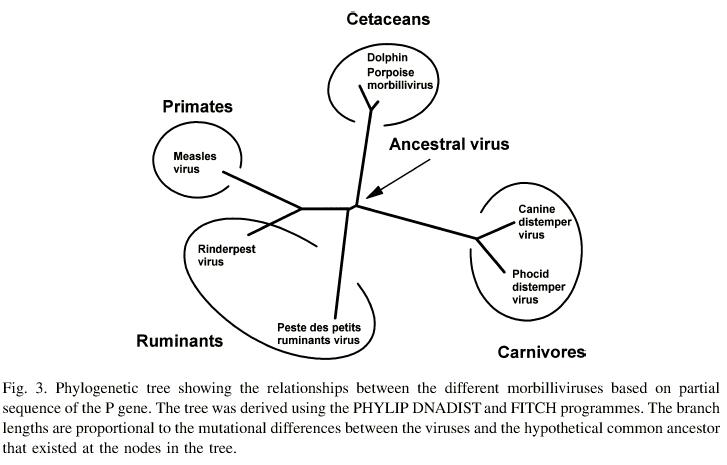
- at an intermediate level, the (apparently) more recent origin
of a variety of different strains of marine mammal viruses,
probably from CDV
(these viruses may also be traded between different
marine mammal populations, or persistent within some
particularly large populations). Molecular data
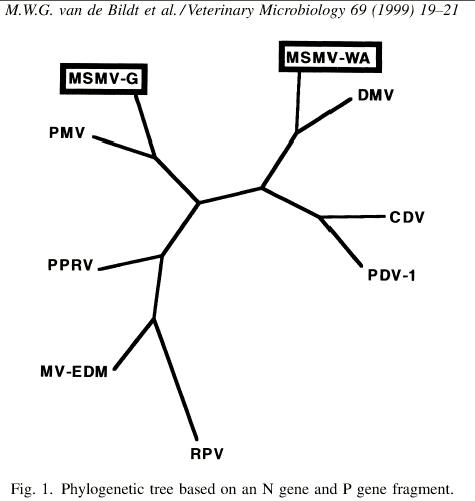
(RPV=rinderpest; PDV=phocine distemper virus; DMV=dolphin
morbillivirus;
CDV=canine distemper virus; PMV=porpoise morbillivirus;
PPRV=peste des petits ruminants;
MSMV=monk seal morbillivirus (WA, West Africa,
and G, greece); MV-EDM=measles virus, Edmonton (vaccine)
strain)
- canine distemper virus seems to have very few limits,
although there are recognizable differences between
strains; however, these strains are mostly geographical
rather than grouped by species, e.g.:
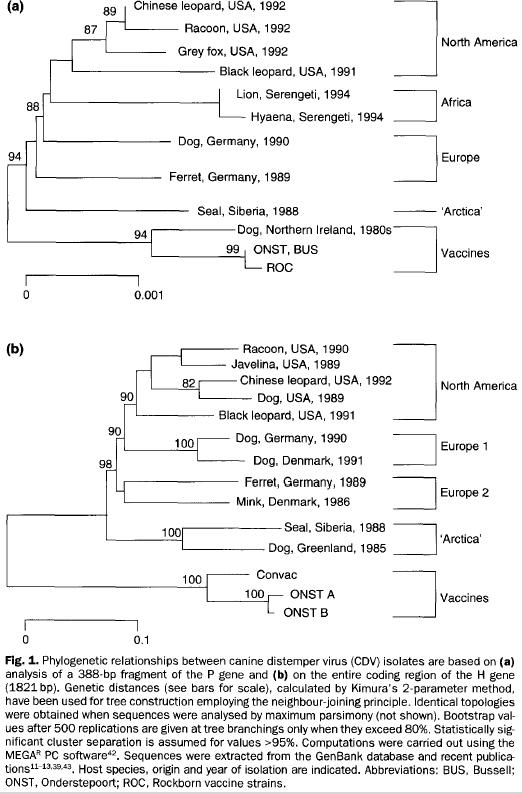
Evolutionary ideas
Morbilliviruses form a "global web" of disease.
The web is dynamic; new strains seem to be forming all the time,
from measles (which apparently evolved in the last 10,000 years),
to the closely related marine mammal family, to the related
strains in CDV epidemics.
If we are sufficiently careful we can draw some
analogies between metazoan parasites in a single host
(when the host dies the parasite has to move on),
morbilliviruses in a population, and maybe even in the long run
diseases in a population that goes extinct (although there's a problem
here, since we are unlikely ever to know about entire species that
were driven extinct by diseases [although the golden tree
frog in Costa Rica -- driven to disappearance
and possible extinction by a chytrid fungus -- may, alas, be an example]).


